
by SumaLatam | Oct 14, 2025 | Blog
Introduction When working with clinical or health content, translation and privacy must go together. Protecting sensitive data is legally required and essential to maintain patient trust. This guide outlines practical steps to reduce privacy risks across the...

by SumaLatam | Oct 11, 2025 | Blog
Introduction Multicenter trial localization is more than translation. It means harmonizing terminology, synchronizing versions, and ensuring collected data are comparable across sites and countries. Clear processes reduce regulatory risk, boost data quality and speed...

by SumaLatam | Oct 9, 2025 | Blog
Introduction Hiring medical translation vendors requires more than price comparison. Regulated content demands accuracy, traceability and subject-matter expertise. This practical guide covers selection criteria, how to build an RFP, sample tests to request and...
by SumaLatam | Oct 7, 2025 | Blog
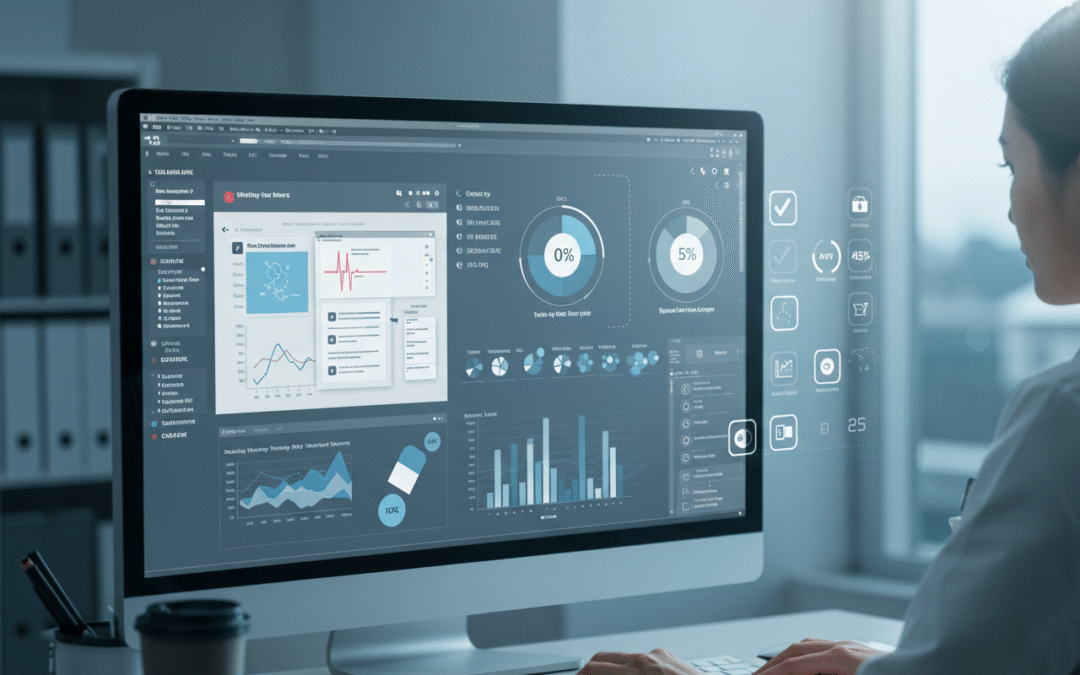
Introduction Measuring medical translation quality is vital to ensure safety, compliance and trust. This guide lists actionable KPIs —from accuracy to turnaround time— and shows how to display them in a QA dashboard. Use these metrics to cut risk and rework. Key KPIs...

by SumaLatam | Sep 20, 2025 | Blog
Medical translation is not only about linguistic precision. In a heavily regulated field like healthcare, every translated document must comply with country-specific and content-specific requirements. Overlooking these rules can lead to approval delays, rejections, or...

by SumaLatam | Sep 18, 2025 | Blog
In medical translation, accuracy is not only about choosing the right word — it’s also about ensuring consistency in medical terminology. In projects where multiple teams or vendors translate medical content, lack of uniformity can cause serious confusion for both...







